Clinical Trials BC celebrates International Clinical Trials Day: recognition of the largest clinical trial ever conducted
21 May 2022
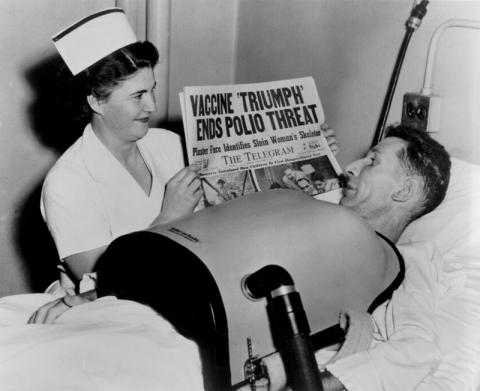
Photo: Nurse with a Toronto Telegram newspaper reporting the April 12, 1955 results of the Salk polio vaccine field trial; the man affected by polio is using a portable respirator.[vii]
May 20 is celebrated every year to mark James Lind’s first controlled clinical trial using citrus fruit on a group of sailors suffering from scurvy.
In 2021, Clinical Trials BC spotlighted the development of the polio vaccine that led to the largest Phase III and Phase IV clinical trials ever conducted, set the stage for successful global immunization programs, and influenced our modern-day vaccine successes. Canada played a huge part in the development and conduct of these trials and related research.
What is Polio?
Polio (poliomyelitis) is an infectious disease that has existed since ancient times and is caused by a virus. It occurs most commonly in children and young adults and can result in paralysis. The disease reached epidemic proportions throughout the first half of the 20th century. Polio inspired fear because it struck without warning, and researchers were unsure how it spread from person to person. Most sources of polio are through the contaminated water supply. The virus can attack the nervous system. Sadly, once contracted, there is no cure; therefore, the prevention of this disease is vital.
During the first half of the 20th century, polio (poliomyelitis), a.k.a. “The Crippler,” hit Canada harder than anywhere else globally. A succession of polio epidemics struck a national crisis between 1946 and 1953 when it became the most severe, reaching every province and even parts of the Arctic.
Scientists at Connaught Medical Research Laboratories at the University of Toronto made a critical ingredient that enabled American medical researcher and virologist Dr. Jonas Salk to prepare the first polio vaccine. The development of Medium 199 from Canada sped up the work. Salk requested a supply of Medium 199, with which he prepared an inactivated poliovirus vaccine (IPV) that could be safely tested on children. Connaught Labs also to provided the poliovirus fluids for the large-scale field trials of the vaccine.[i – all sources cited at the bottom of this page]
On April 26, 1954, the Salk polio vaccine field clinical trials involving 1.8 million children began. Children in Canada, the USA, and Finland participated in the trials, which used for the first time the now-standard double-blind clinical trial method, whereby neither the patient nor attending doctor knew if the inoculation was the vaccine or a placebo.[ii] Participants in the polio clinical trials were called “polio pioneers.” [iii]
The largest and most expensive phase III Clinical Trials ever conducted were in 1954 and 1959, involving 1.8 million participants in North America and 10 million in Russia, known as the Polio Field Trials!

Photo: Dr. Jonas Salk inoculating a polio clinical trial participant, a ‘polio pioneer,’ with the investigational vaccine [iv]
One year later, on April 12, 1955, researchers announced that the polio vaccine was safe and effective. News of the successful vaccine came via radio announcements, headlines in newspapers, and church bells ringing. Children were let out of school, and people danced and embraced in the streets. [v] There was good statistical evidence that Jonas Salk’s killed-virus preparation was 80-90% effective in preventing paralytic poliomyelitis.[vi] Salk’s vaccine quickly became a standard part of pediatric immunization in North America.
A decision was made in Canada to proceed with the immunization of Canadians with the Connaught prepared Salk vaccine, which was the only one used in Canada. Canada immediately implemented Phase IV surveillance (trials and evaluation) of the vaccine showing that it was safe and effective. This helped pave the way for other countries, including the USA, to start immunization.
Connaught Labs at UofT also solved the problem of producing the vaccine on a large scale and immediately took over polio vaccine production and distribution, which resulted in an abundant supply of the Salk vaccine and an inoculation campaign for all youth in North America. There was enough to export to countries that had not yet produced their own.
Next came the Sabin oral polio vaccine in 1959., Dr. Albert Sabin accepted Russia’s invitation to conduct a massive trial of his oral vaccine on 10 million Russian children. The oral polio trial was a success, and Russia immediately ordered the vaccination of every person under the age of 20 with Sabin’s vaccine, and an estimated total of 77 million people were vaccinated.
In the following decades, polio vaccines would irradicate the highly contagious disease in the Western Hemisphere and most developed countries.
Rotary International has been working to eradicate polio since 1979. In 1987 The WHO identified pockets of the globe where the wild poliovirus still ran largely unchecked. With its founding partner Rotary International, the WHO launched it’s Global Polio Eradication Initiative (GPEI) to target endemic polio.
Thanks to both the Salk and Sabin clinical trials and vaccines and the efforts of Rotary International and its expanded partners of the GPEI, infections from the wild-type poliovirus have been eradicated worldwide in all but two countries. Polio remains endemic only in Afghanistan and Pakistan, which reported 176 new infections in 2019.
Rotary International has contributed extensive funding, developed trusting relationships with communities and set up robust infrastructure for immunization in 122 countries. Along with its partners, they have helped immunize more than 2.5 billion children against polio.[viii]
It is crucial to continue working to keep countries polio-free. If all eradication efforts stopped today, polio could paralyze as many as 200,000 children each year within ten years.[ix]
“Covid-19 reminds us what can happen if we were to stop ongoing vigilance and surveillance studies.” – Tania Hall, Rotarian, Sunshine Coast Rotary Club[x]
Rotary clubs worldwide have stepped forward and are now mobilizing in their communities to support the introduction and delivery of COVID-19 vaccinations. The infrastructure created by Rotary International and its Global Polio Eradication Initiative (GPEI) partners is already supporting efforts to combat COVID-19. The GPEI supports the rollout of COVID-19 vaccines. Rotary is active at the local level, promoting community education, ensuring equitable distribution of the vaccine, helping stop the continued spread of the disease, aiding local health authorities to prepare for mass vaccinations and enabling surveillance trials.
After vaccine clinical trials are deemed successful, the world still faces challenges: Continuing surveillance trials and equal distribution of available vaccine for the rest of the world.
‘Modern research and clinical trials are not conducted by lone scientists like Jonas Salk or Albert Sabin, but by huge teams of people all working together. Clinical Trials BC would like to thank and celebrate the success of clinical trials and the vaccination programs with the researchers, lab personnel, research techs, regulatory personnel and reviewers, qualified investigators, clinical trials research teams, pharmaceutical companies, and a host of dedicated volunteers from organizations determined to immunize the world.[xi]
References and Acknowledgements:
[i] Canada and the Development of the Polio Vaccine | The Canadian Encyclopedia
[ii] Polio Vaccine Trials Begin – HISTORY
[iii] 8 Things You May Not Know About Jonas Salk and the Polio Vaccine – HISTORY Section 5.
[iv] Photo Credit: Al Fenn/The LIFE Picture Collection/Getty Images: download May 11, 2021.
[v] How dimes defeated polio – Chicago Tribune
[vi] Francis T, Jr, Korns R, Voight R, Boisen M, Hemphill F, Napier J, et al. An evaluation of the 1954 poliomyelitis vaccine trials: summary report. Am J Public Health. 1955;45(suppl):1–50. [PMC free article] [PubMed] [Google Scholar] download May 11, 2021
[vii] Photo source is form the March of Dimes Canada as published in the article Canada and the Development of the Polio Vaccine | The Canadian Encyclopedia
[viii] Ending Polio | Rotary International
[ix] Ending Polio | Rotary International
[x] Quote from Tania Hall, Rotarian, Sunshine Coast Rotary Club, British Columbia, Canada, Rotary International District 5040
[xi] Quote from Clinical Trials BC on the occasion of International Clinical Trials Day 2021 – Alison Orth, Jean Smart, Marc Saunders, Brianna Stowell, Dan Enjo.
Acknowledgements & Guiding Principles





